Diffusion of Ammonia and HCl.
• C+55+35
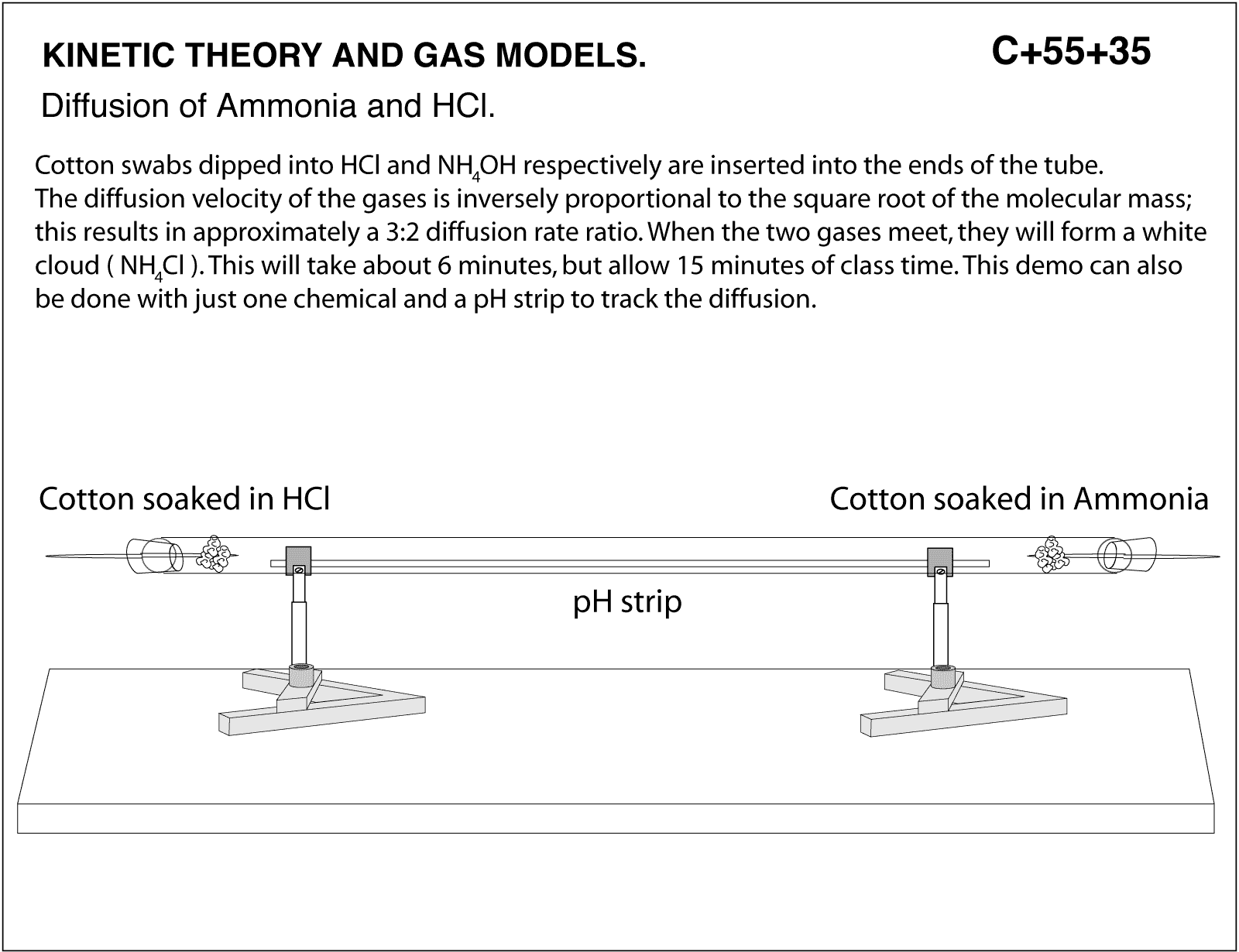
Diffusion of Ammonia and HCl. Cotton swabs dipped into HCl and NH40H respectively are inserted into the ends of the tube. The diffusion velocity of the gases is inversely proportional to the square root of the molecular mass; this results in approximately 3:2 diffusion rate ratio. When the two gases meet, tey will form a white cloud (NH4Cl). this will take about 6 minutes, but allow 15 minutes of class time. This demo can also be done with just one chemical and a pH strip to track the diffusion.

Related Demos
C+55+0
Mechanical model of a gas: Vibrating balls on OHP. C+55+5
Hexstat probability device. C+55+10
Mechanical model of a gas: Vibrating balls strike piston on OHP. C+55+15
Brownian motion: Smoke particles viewed using TV camera. C+55+20
Brownian motion: Like C+55+0, with an aluminum disk added. C+55+25
Stoekle's apparatus: Heated tube with mercury and glass bits. C+55+30
Ruchardt's tube: Ball oscillates in a vertical glass tube on jug. C+55+40
Osmosis of helium through semi-porous cup. C+55+45
Viscosity of air: One rotating disk drives another. C+55+55
Breathe helium, voice pitch rises; breathe SF6, voice pitch lowers. C+55+60
Equipartition of Energy: Different mass balls bounce out at different times. C+55+65
Applet: Gas Properties C+55+70
Applet: A molecular explanation of pressure C+55+75
Applet: Brownian motion C+55+85
Applet: Maxwell's Demon